Abstract
Enzyme activity and biochemical constituents of healthy and stem rot infected tissues of susceptible variety of groundnut TMV2 were evaluated. Results revealed that higher activity of Oxidative enzymes (catalase, polyphenol oxidase and peroxidase) were present in infected leaves than healthy ones. In case of Biochemical constituents, higher amount of total and Ortho-di hydric phenols and protein content was observed in infected plants as compared to healthy plants. Higher content of sugars and chlorophyll was observed in healthy plants as compared to infected plants.
Introduction
Groundnut (Arachis hypogaea L.) is an important annual oil seed crop4. A large number of diseases attack groundnut in India15167. The majority is caused by fungi and several of them are yield reducers in different regions and seasons17. Among the soil-borne fungal diseases, stem rot caused by S.rolfsii is a potential threat to groundnut production and is of considerable economic significance for the crop grown under irrigated conditions. This disease causes severe damage during any stage of growth, and yield losses over 25% have been reported16. The present communication deals with the role of various oxidative enzymes and different Biochemical constituents in imparting resistance to groundnut plants against stem rot disease.
Materials and methods
Enzyme assay:
Healthy and infected seedlings of TMV2 variety were taken and different enzymes viz.catalase, peroxidase and polyphenol oxidase were assayed at 5 different stages (1, 3, 5, 7 and 11 days) to know about their role in defense mechanism of groundnut plants against stem rot disease. Catalase, peroxidase and polyphenol oxidase were assayed as per Thimmaiah26. Catalase activity was assayed by estimating the residual H2O2 in the reaction mixture which was then determined by oxidation with KMnO4 titrimetrically. The catalase activity was expressed as mg of hydrogen peroxide destroyed in five min per gram fresh weight of the tissue. Peroxidase activity was assayed using o-dianisidine as hydrogen donor and H2O2 as electron acceptor. The rate of formation of yellow orange colored dianisidine dehydrogenation product was a measure of the peroxidase activity and assayed spectrophotometrically at 430 nm. The specific activity of enzyme was expressed as units/min/mg protein. Polyphenol oxidase was measured as rate of increase in absorbance colorimetrically at 410 nm with the oxidation of catechol as the substrate. The enzymatic activity was expressed as units min.-1 at 410 nm considering one enzyme unit as the change in absorbance of 0.001/minute and the specific activity as units/min/mg protein.
Biochemical constituents:
For assaying biochemical constituents healthy and infected tissues of TMV2 variety of groundnut were analyzed for contents of various biochemical constituents viz. total sugars, reducing sugars, non-reducing sugars, total and ortho-dihydric phenols, soluble protein and chlorophyll to know their possible role in imparting disease resistance. For estimation of sugars and phenols seedlings of 5 stages (1, 3, 5, 7 and 11 days) both healthy and diseased were used. Ethanol extraction of plant material was made by following the method of Mahadevan et al.,11. Ethanol extraction was used for estimating phenols and sugars. Reducing sugars were estimated by method of Nelson18, total sugars was calculated by the method of Inman8 and Nelson18. And non-reducing sugars were calculated by subtracting the reducing sugars from total sugar content. Total phenols were estimated as per Bray and Thorpe3. Ortho-di hydric phenols were estimated as per Mahadevan and Sridhar12. The estimation of proteins was done as per method developed by Lowry et al.,10. Estimation of chlorophyll was done as per Thimmaiah26.
Results and Discussion
An insight in to the data (Table 1) showed that diseased samples showed higher enzyme activity than their healthy samples and with increase in age of plants the enzyme activity also showed in increasing trend. Our results are in agreement with the findings of Rajalaxmi and Ramarethinam20 and Avallone et al.,1 who reported increased activity of polyphenol oxidase and peroxidase in tea leaves infected with blister and increased activity of laccase in pine apple fruits infected by black spot.
Table 1: Activity of different oxidative enzymes in healthy and infected groundnut hypocotyls at various stages of disease development. Each value is an average of 3 replicate samples ± Standard error (S.E) + = Increase over control.
Different enzymes have different mode of action in imparting resistance. Increased peroxidase activity has often been studied in connection with the oxidation of phenolic substances in the diseased plants and resistance in host attributed to toxicity of these oxidation products5. Moreover, enhanced peroxidase activity has been linked with synthesis of lignin22. Catalase activity increased during infection as a mechanism to scavange fungitoxic H2O2 (Montalbini, 1991). polyphenol oxidase is commonly found in plant. It oxidizes phenols to highly fungitoxic quinines and is highly inhibitory to some plant fungi30. Sain and Gour23 showed increased levels of peroxidase and catalase in Parthenium hysterophorus infected with X.axonopodis. In our studies the increased enzyme activity can be attributed to activation, solubilization or de novo synthesis of enzymes or their products by the invading pathogen.
Biochemical resistance or susceptibility in plants against any disease depends mainly on pre-existing; pre formed or induced substances by the pathogen in the host. The nutritional status and concentration of biochemical constituents in plants prior to infection may determine the severity of disease. Sugars play an active role in the inhibition of pectinolytic and cellulolytic enzymes which are essential for the pathogen2. Moreover, sugars are precursors of phenolics which are highly toxic to microorganisms (Vidyasekharan, 1978). Healthy plants recorded higher amount of sugars than diseased plants (Table 2). Our results are in agreement with the findings of Verma and Singh28 and Sultana et al.,25 who reported higher amount of sugars in healthy plant parts as compared to diseased ones. The reduction in sugar content after infection may be due to rapid hydrolysis of sugars during pathogenesis through enzymes (hydrolases)secreted by pathogens and subsequent utilization by pathogens for their development.
Table 2: Sugars (mg/gm fresh weight) in healthy and infected groundnut hypocotyls at various stages of disease development. Each value is an average of 3 replicate samples ± Standard error (S.E); - = Decrease over control.
In our studies, higher amount of total and O.D. phenols was observed in diseased plants as compared to healthy plants (Table 3). The results of present studies are in agreement with the findings of Ganguly6 and Singh24 who observed higher phenol content in diseased plant parts of resistant varieties.
The post-infection increase in phenolic content could be due to a number of factors including enhancement of synthesis, translocation of phenolics to the site of infection and hydrolysis of phenolic glycosides by fungal glycosidases to yield free phenols and the increase in level of phenolic compounds in infected leaves may be due to translocation of phenolics to the site of infection19.
Table 3: Total and ortho-dihydric phenols (mg/gm fresh weight) in healthy and infected groundnut hypocotyls at various stages of disease development. Each value is an average of 3 replicate samples ± Standard error (S.E); + = Increase over control.
Analysis of protein content revealed higher protein content in diseased plants as compared to healthy plants (Table 4). Kaur and Dhillon9 observed an increase in protein content of groundnut infected with Cercospora personatum. Higher total protein content in infected pods of T-9 variety of Vigna mungo susceptible to leaf crinkle virus was observed by Malik et al.,13. It is a well-known fact that enzymes are proteins and the increased synthesis of proteins during the infection may be due to activation of enzymes which are essential for the synthesis of various defense chemicals31. The increase in protein content may have resulted from synthesized proteins in the infected host27.
Table 4: Total proteins (mg/gm fresh weight) in healthy and infected groundnut hypocotyls at various stages of disease development. Each value is an average of 3 replicate samples ± Standard error (S.E); + = Increase over control.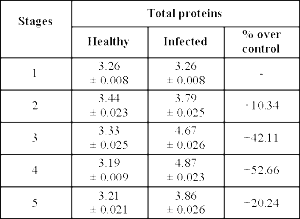
Investigations on chlorophyll content revealed that higher content was observed in healthy plants as compared to diseased plant parts (Table 5). The loss of chlorophyll in diseased leaves under the present investigation may be due to interference by pathogen in the normal chlorophyll synthesis or breakdown of chlorophyll due to activation of enzymes that degrade chlorophyll21.
Table 5: Chlorophyll (mg/gm fresh weight) in healthy and infected groundnut hypocotyls at various stages of disease development. Each value is an average of 3 replicate samples ± Standard error (S.E); - = Decrease over control.
Acknowledgements
This work forms part of program of work of the Major Research Project sanctioned to Prof M N Reddy, by the University Grants Commission, New Delhi, India. Thanks are also due to the sanction of Fellowship to the Project Fellow, Sireesha, CH in the Project.References
- Avallone S, Guiranol JP, Brilliant JM and Teisson C. 2003. Enzymic browning and biochemical alterations in black spot of pineapple. Curr. Sci., 47:113-118.
- Bateman DF and Millar RL. 1966. Pectic enzymes in tissue degradation. Ann. Rev. phytopath., 4:119-146.
- Bray HG and Thorpe WV. 1954. Analysis of phenolic compounds of interest in metabolism. Methods of Biochemical analysis 1:27-52.
- Brown RG. 1999. Diseases of Cereal Crops and Annual Oil Seed Crops. In: Plant Diseases and their Control. (Ed. R.G. Brown). Sarup & Sons, New Delhi. 297-331.
- Fric F. 1976. Oxidative enzymes. In: Encyclopedia of plant physiology-IV. (Ed.) R. Heitefuss and P.H. Williams. Physiological Plant Pathology. Springer-Verlag, Berlin, Heidelberg. New York. 617-631.
- Ganguly LK. 1995. Biochemical alteration in respect of total phenols, sugars and protein contents in Rhizoctonia solani Kuhn. Inoculated rice plants. J. Mycopathol. Res., 33:53-58.
- Ganesan S and Sekar R. 2004a. Biocontrol mechanism of Groundnut (Arachis hypogaea L.) Diseases-Trichoderma system. In: Biotechnological applications in Environment and Agriculture, (Eds.G.R.Pathade and P.K. Goel).ABD Pub. Jaipur. India. 312-327.
- Inman RE. 1962. Disease development, disease intensity and carbohydrate levels in rusted bean plants. Phytopathology 52:1207-1211.
- Kaur J and Dhillon M. 1990. Biochemical alteration in groundnut leaves induced by Cercosporium personatum. Indian J. mycol. Pl. path. 19:151-156.
- Lowry OH, Rosebrough NJ, Farr AL and Randall RJ. 1951. Protein measurement with the Folin-Phenol reagent. J. Biol. Chem., 193:265-275.
- Mahadevan A, Kuc J and Williams EB. 1965. Phytopathology 55:1000-1003.
- Mahadevan A and Sridhar R. 1986. Methods in physiological plant pathology. (3rd Ed.) Sivakami Publications, Chennai. 183-184.
- Malik S, Kumar P, Panwar JDS and Rathi YPS. 2002. Physiological and biochemical alterations induced by urdbean leaf crinkle virus in Vigna mungo. Ann. Pl. protect. Sci., 10:91-94.
- Montalibini P. 1991. Effect of rust infection on leaves of uricase allantoinase and ureides in susceptible and hypersensitive bean leaves. Physiol. Mol. Pl. Pathol., 39:173-188.
- Mayee CD. 1987. Diseases of groundnut and their management. In: plant protection in field crops. (Eds., M.V.N. Rao and S.Sitanantham).PPSI, Hyderabad. 235-243.
- Mayee CD and Datur VV. 1988. Diseases of groundnut in the tropics. Review Trop. Pl. path., 5:169-198.
- Mayee CD. 1995. Current status and future approaches for management of groundnut disease in india. indian phytopath., 48:389-401.
- Nelson, N. 1944. A photometric adoption of the somogyi method for the determination of Gencose. J. biol. Chem., 153:375-380.
- Parashar RD, Sindhan GS and Hooda l. 1987. Role of phenolic compounds and carbohydrates in resistance of bajra cultivars to downy mildew. Pl. dis. 2:120-121.
- Raja Lakshmi M and Ramarethinam S. 2000. The role of exobasidium vexans in flavonoid synthesis by Camellia assamica. J. Plant. Crops 28:357-359.
- Rathore BS, Pathak VN and Rathore RS. 2001. Impact of downy mildew on bio constituents and rate of transpiration in Blond-psyllium plants. J. Mycol. Pl. pathol., 31:258-260.
- Ride JP. 1975. Lignification of wounded wheat leaves in response to fungi and its possible role in resistance. Physiol., Plant pathology 5:125-134.
- Sain SK and Gour HN. 2008. pathological, physiological and biochemical characterization of xanthomonas axonopodis pv.parthenii incident of leaf blight in Parthenium hysterophorus.J. Mycol. Pl. Pathol., 38:466-477.
- Singh HV. 2000. biochemical basis of resistance in Brassica species against downy mildew and white rust of mustard. Pl. dis. Res., 15:75-77.
- Sultana N, Kasem MA, Hussain MD and Alam MS. 1998. Biochemical changes of some promising lines of yard long bean due to infection of yellow mosaic virus. Thai J. agric. Sci., 31:322-327.
- Thimmaiah SK. 1999. Standard methods of biochemical analysis. Kalyani publishers 184-253.
- Uritani l. 1971. Protein changes in diseased plants. Ann. Rev. Phytopath., 9:211-234.
- Verma KS and Singh T. 1994. biochemical alterations in grey blight affected leaves of mango. Pl. dis. Res., 9:29-34.
- Vidyasekaran P. 1978. Production of pectinolytic enzymes by Helminthosporium nodulosum in vitro and in vivo. Indian phytopath., 28:451.
- Vidyasekaran P. 1993. Principles of plant pathology. C.B.S. publishers and distributers. New Delhi. 83-92.
- Vidyasekaran P. 2001. Physiology of disease resistance. In: principles of plant pathology (1st Ed.). C.B.S. publishers & distributers. New Delhi. 106-116.